COPD Guidelines: The COPD-X plan
COPD-X V2.77, December 2024
Lung Foundation Australia’s COPD Guidelines Committee, manages the co-branded Lung Foundation and Thoracic Society of Australia and New Zealand’s, “The COPD-X Plan: Australian and New Zealand guidelines for the management of chronic obstructive pulmonary disease”. To ensure you stay up-to-date with these guidelines, register below.
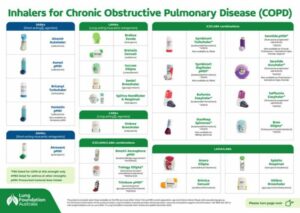
COPD-X Handbook
Updated 2024: Our most popular point-of-care COPD-X summary is now in an easy-to-navigate, mobile-friendly format. Find the latest practice points and supporting information links most requested by healthcare professionals across primary care, specialist, and hospital settings.
Download
COPD-X is updated every quarter. Register here to receive free updates of the latest evidence in COPD